Sermorelin 10mg Supply for Clinics
Growth Hormone Peptide: Sermorelin Clinical Supply
Sermorelin Acetate 10mg for Clinical Supply
Injecta Connect facilitates the sourcing and procurement of Sermorelin Acetate (10mg) on behalf of licensed medical practices, clinics, hospitals, and authorized healthcare institutions from licensed compounding pharmacies, authorized wholesalers, and verified distributors.
Sermorelin is a synthetic analog of growth hormone-releasing hormone (GHRH) that may be prescribed by licensed providers within appropriate clinical contexts to stimulate endogenous growth hormone (GH) secretion.
Injecta Connect does not prescribe, dispense, compound, or provide medical advice. Clinical decisions regarding use, dosing, indications, and patient suitability remain solely the responsibility of the prescribing provider.
Product Overview
- Generic Name: Sermorelin Acetate
- Presentation: Lyophilized powder for reconstitution and subcutaneous injection
- Strength: 10mg per vial
- Category: Growth Hormone-Releasing Hormone (GHRH) Analog (Peptide Therapy)
- Rx Status: Prescription-only medication. All orders require valid licensing and provider credentials.

The Injecta Connect Advantage
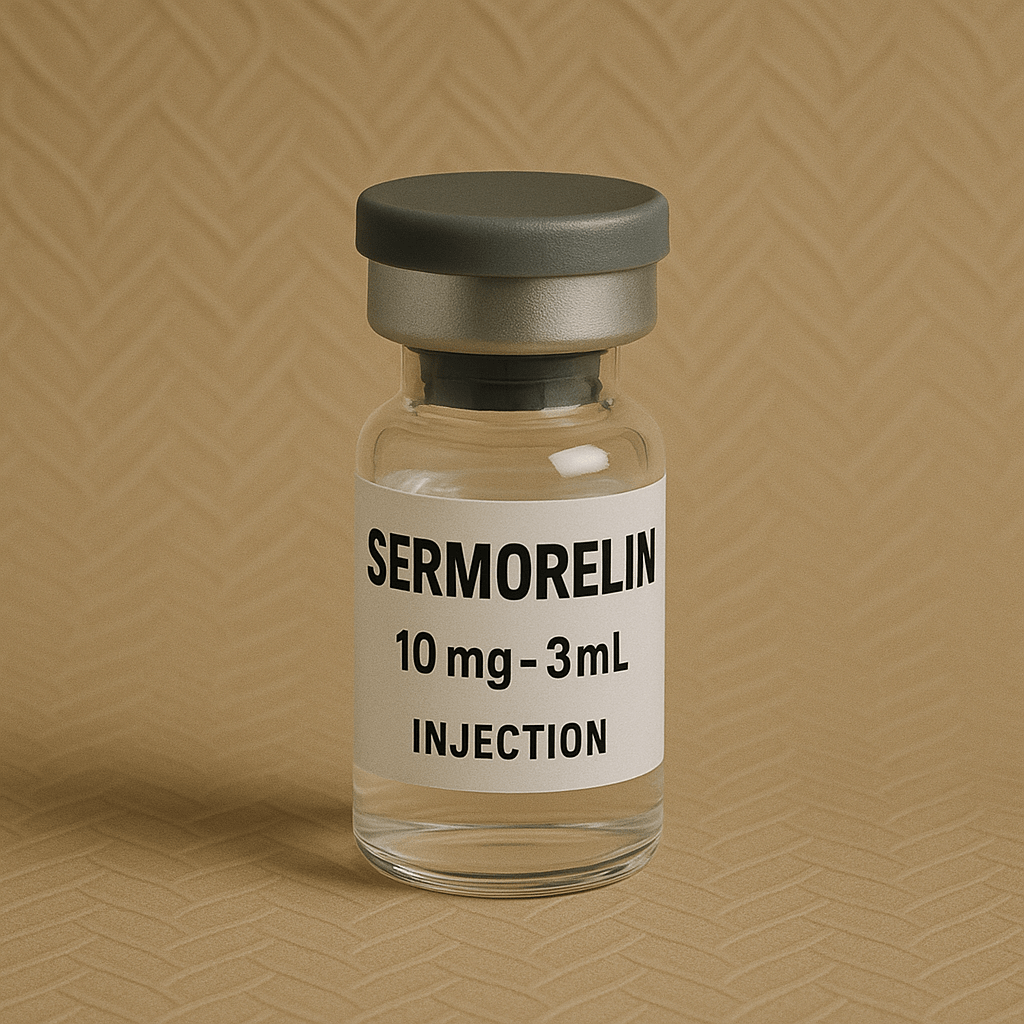
- Available in stock
Why Licensed Providers Use Injecta Connect
- Streamlined procurement coordination
- Access to verified pharmacies and authorized distributors
- Transparent sourcing pathways
- No patient-facing sales model
- B2B-only registration and verification
We serve licensed medical entities seeking compliant supply chain support (not direct-to-consumer buyers)

Sermorelin Clinical Context

Sermorelin is commonly utilized by licensed practitioners in the following therapeutic areas such as:
- Growth hormone deficiency assessment and support
- Age-related hormonal optimization
- Functional medicine and integrative wellness protocols
- Body composition management
- Sleep architecture improvement
- Cellular repair and recovery support
This information is intended as a product reference for licensed healthcare professionals already familiar with Sermorelin’s pharmacology and clinical applications. It does not constitute prescribing guidance.
All treatment decisions must be made by the prescribing provider based on individual patient assessment.

Regulatory & Compliance Framework

Injecta Connect operates strictly as a procurement coordination platform for Sermorelin clinical supply.
We do not:
- Advertise to consumers
- Dispense medications
- Provide treatment protocols
- Offer dosing guidance
- Guarantee therapeutic outcomes
All medications are supplied through properly licensed entities in accordance with:
- U.S. Food and Drug Administration regulatory requirements
- Applicable state board of pharmacy regulations
- Federal compounding standards
Healthcare providers are responsible for ensuring compliance within their jurisdiction.
Disclaimer:
Product information provided on this page is intended for licensed healthcare professionals and is not intended as patient education, marketing to consumers, or prescribing guidance. Sermorelin Acetate is a prescription medication. Federal law restricts this drug to use by or on the order of a licensed healthcare provider.
Injecta Connect does not take physical possession of, repackage, relabel, compound, or dispense any pharmaceutical product. All trademarks and product names referenced are the property of their respective owners and are used for identification purposes only.

Sermorelin 10mg Sourcing
Frequently Asked Questions
Who can place an order for Sermorelin 10mg through Injecta Connect?
Only verified licensed physicians, clinics, hospitals, and regulated healthcare institutions may request procurement support.
What is Injecta Connect’s role in the Sermorelin supply process?
Injecta Connect acts solely as a B2B procurement coordination platform and does not prescribe, dispense, or compound medications.
How is regulatory compliance maintained in the supply chain?
All sourcing is coordinated through licensed pharmacies or authorized distributors in accordance with U.S. Food and Drug Administration regulations and applicable state laws.
Who determines clinical protocols, dosing, and patient eligibility?
All clinical decisions remain exclusively under the authority and responsibility of the licensed prescribing provider.
